GIT Transit Time & SIBO: Accurate Testing Starts with Timing
What is GI transit time? Gastrointestinal (GI) transit time is the duration required for ingested material to pass through the digestive tract from...
4 min read
![]() Dr. Andrea Gruszecki, ND
:
November 15, 2022 at 12:44 PM
Dr. Andrea Gruszecki, ND
:
November 15, 2022 at 12:44 PM

When a patient reports a significant improvement, it can be tempting for clinicians to ignore follow-up testing. Testing is expensive and may require dietary changes, blood draws, or sample collections that can disrupt daily routines or be challenging for patients.
Follow-up testing to document improvement is not only a best medical practice but on occasion, it can modify the final diagnosis or further adjust a nutritional protocol. Recently, I consulted with a clinician on pre/post Organic Acids (OAPs) regarding a 6-year-old child with developmental delays who do not use any pharmaceutical medications.
The patient’s initial presentation included:
The clinician immediately ordered food sensitivity testing and then the Organic Acids profile. While waiting for the OAP results to come back the patient discontinued wheat, corn, and dairy based on the food sensitivity results. The clinician also increased healthy fats and hydration. The food eliminations and diet changes significantly improved constipation and defecation but had little effect on the apraxia.
The OAP results indicated significant dysregulation of the patient’s biochemical and mitochondrial function. Dysregulation of biochemistry and/or mitochondrial function can result from an inheritance, environmental exposures, psychological stress, or poor nutrition. Biochemical pathways can be overwhelmed by too many calories (over-nutrition) or starved if the proper nutrient cofactors are unavailable in the diet. Inflammation and dysregulation result from either dietary choice and the dysregulation is reflected in the OAP results. An Environmental Pollutants profile (EPP) collected simultaneously with the Organic Acids profile indicated recent exposures to styrene, phthalates, parabens, xylene, and MTBE (gasoline additive).
A protocol was designed after this first Organic Acids test and the child made significant improvements in speech and movement and started to catch up on developmental milestones. The OAP test indicated additional nutritional supports, including:
The chemical exposures were tracked down to the food served on plastic dishes and eliminated, and the patient was supported with a gentle detoxification protocol. Once these corrections were in place the child began to improve in energy level, and muscle tone (posture). She began speaking in full sentences and started counting. She also learned to write her name and draw recognizable images instead of scribbles.
After a few months of growth and improvement, however, the child plateaued; she began to experience fatigue again in June/July 2022, began finger-sucking, and experienced knee pain when running. At the visit, the mother reports that the patient has been primarily eating carbohydrates.
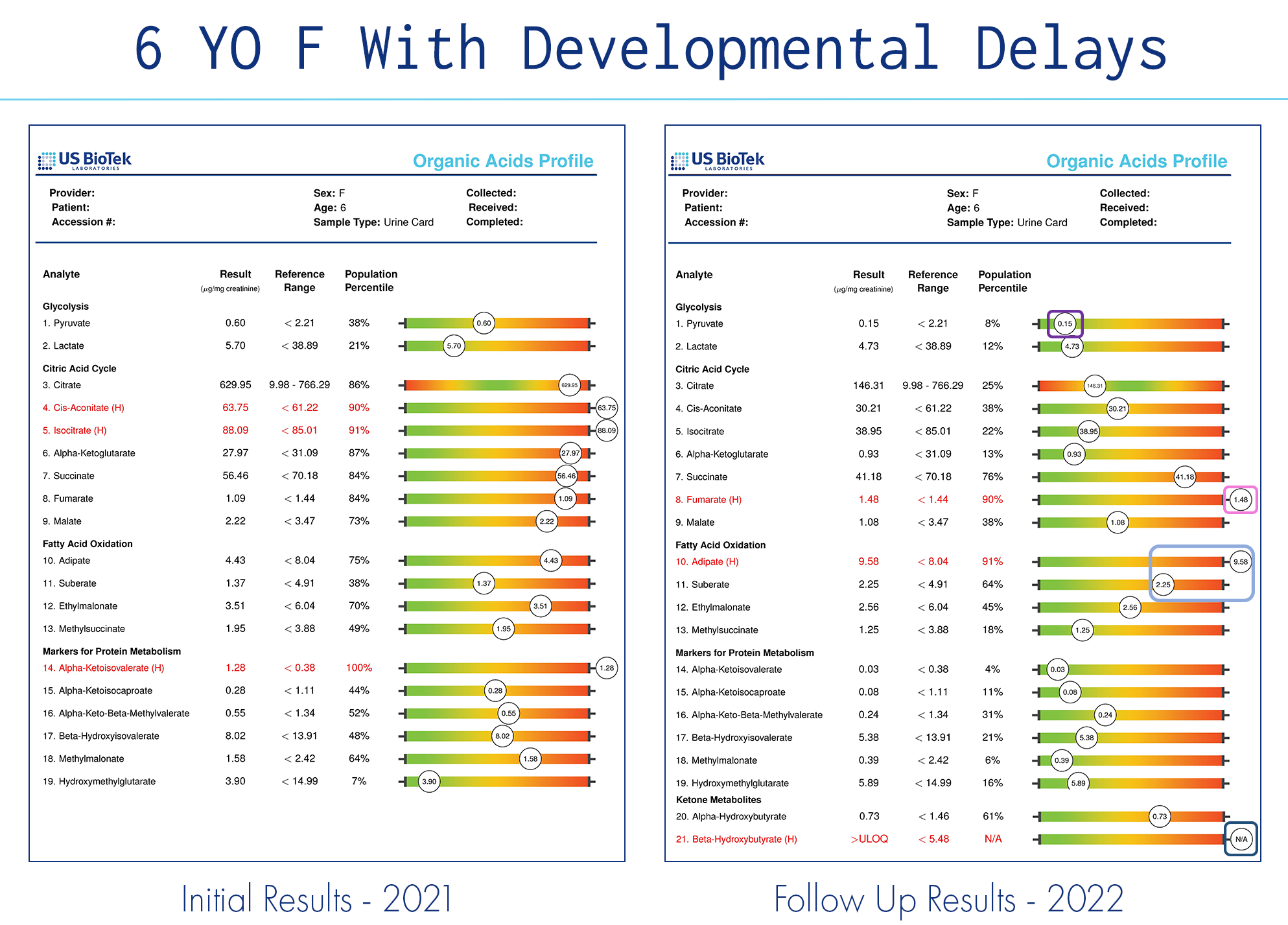
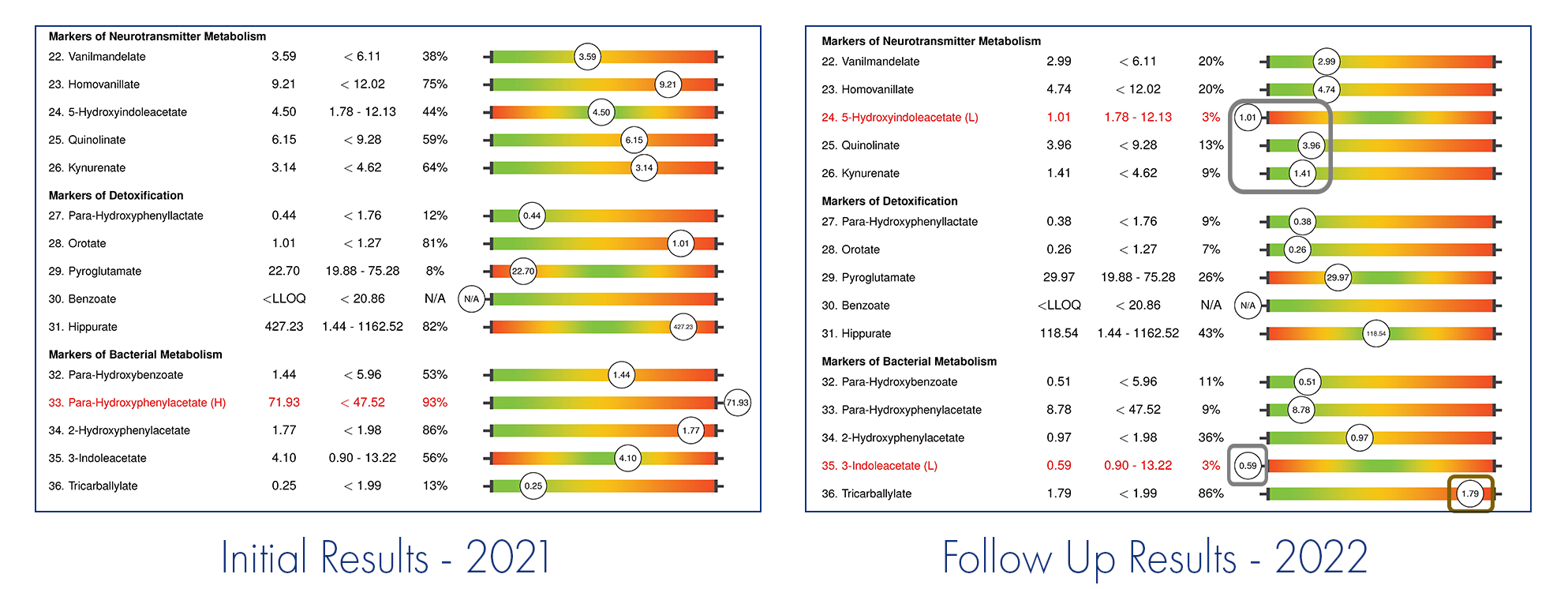
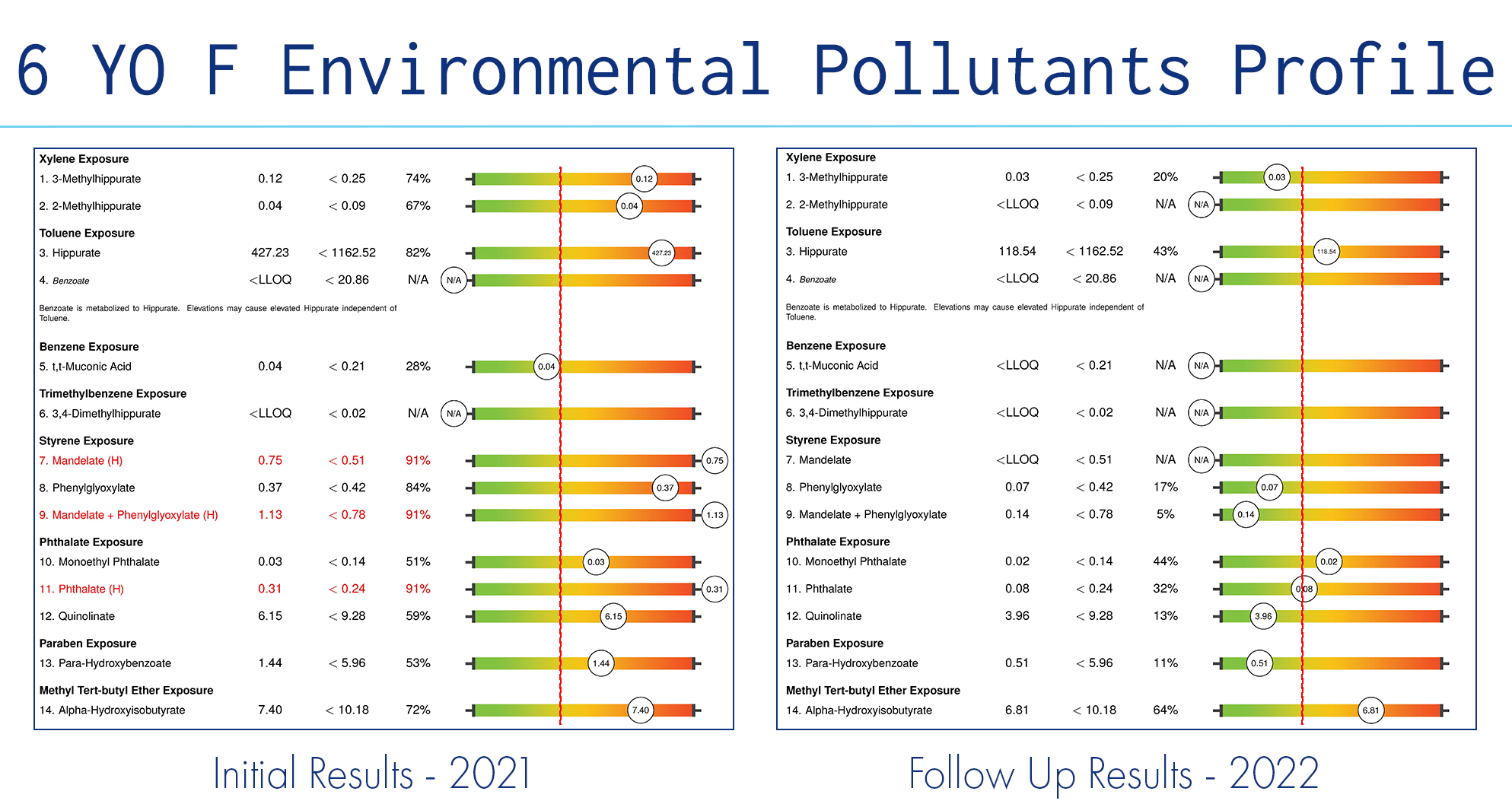
A second OAP was ordered, and the results were significantly different due to the corrective protocol applied after the first results. However, while biochemical and mitochondrial function were improved overall, there were several significant findings that potentially explain the child’s developmental plateau, and another, different, toxic exposure has become apparent. Biochemical findings included:
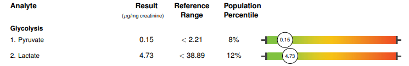
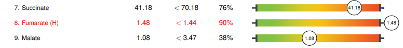
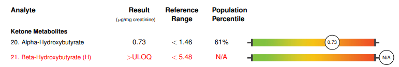
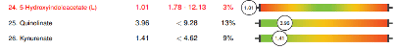
The patient’s protocol was further adjusted and as of October 2022, the mother reports that the patient is now able to keep pace with the family on hikes, is writing her name even better and more legibly, and that the patient’s scoliosis remains stable due to the patient’s improved strength and muscle tone.
Action steps for each of these analytes can be found in US BioTek’s Organic Acids Interpretation guide. Using the information from the guide, can you craft a protocol to support this patient?
Azzolino D, Arosio B, Marzetti E, Calvani R, Cesari M. Nutritional Status as a Mediator of Fatigue and Its Underlying Mechanisms in Older People. Nutrients. 2020 Feb 10;12(2):444.
Chinopoulos C. Which way does the citric acid cycle turn during hypoxia? The critical role of α‐ketoglutarate dehydrogenase complex. J Neurosci Res. 2013;91(8):1030-1043.
Gruszecki A. (2021) Organic Acids Profile and Environmental Pollutants Interpretation Guide. Published by US BioTek Laboratories, Shoreline, WA.
Guthrie L, Wolfson S, Kelly L. The human gut chemical landscape predicts microbe-mediated biotransformation of foods and drugs. Elife. Jun 2019;8:e42866. doi:10.7554/eLife.42866.
Johannsen DL, Ravussin E. The role of mitochondria in health and disease. Curr Opin Pharmacol. 2009;9(6):780-786.
Kumps A, Duez P, Mardens Y. Metabolic, Nutritional, Iatrogenic, and Artifactual Sources of Urinary Organic Acids: A Comprehensive Table. Clin Chem. May 2002;48(5):708–717.
LeMieux M, Al-Jawadi A, Wang S, Moustaid-Moussa N. Metabolic profiling in nutrition and metabolic disorders. Adv Nutr. 2013;4(5):548-550.
Marc DT, Ailts JW, Campeau DC, Bull MJ, Olson KL. Neurotransmitters excreted in the urine as biomarkers of nervous system activity: validity and clinical applicability. Neurosci Biobehav Rev. 2011;35(3):635-644.
Parikh S, Saneto R, Falk MJ, et al. A modern approach to the treatment of mitochondrial disease. Curr Treat Options Neurol. 2009;11(6):414-430.
Pham TX, Lee J. Dietary regulation of histone acetylases and deacetylases for the prevention of metabolic diseases. Nutrients. 2012 Nov 28;4(12):1868-86.
Qiu H, Schlegel V. Impact of nutrient overload on metabolic homeostasis. Nutr Rev. 2018 Sep 1;76(9):693-707.
Ryder B, Moore F, Mitchell A, et al. Fumarase Deficiency: A Safe and Potentially Disease Modifying Effect of High Fat/Low Carbohydrate Diet. JIMD Reports. 2018 ;40:77-83.
Sears ME, Genuis SJ. Environmental determinants of chronic disease and medical approaches: recognition, avoidance, supportive therapy, and detoxification. J Environ Public Health. 2012;2012:356798.

What is GI transit time? Gastrointestinal (GI) transit time is the duration required for ingested material to pass through the digestive tract from...
Overview Many clinicians searching for answers about mycotoxin testing ask questions like “Should I provoke mycotoxins before urine testing?”, “When...

US BioTek Laboratories today announced the completion of its merger with NutriPATH Pathology, a respected functional pathology laboratory serving...