GIT Transit Time & SIBO: Accurate Testing Starts with Timing
What is GI transit time? Gastrointestinal (GI) transit time is the duration required for ingested material to pass through the digestive tract from...
4 min read
![]() Dr. Andrea Gruszecki, ND
:
March 27, 2020 at 1:49 PM
Dr. Andrea Gruszecki, ND
:
March 27, 2020 at 1:49 PM

Every year we run the risk of viral upper respiratory infection by RNA viruses including influenza and common colds (Rhinoviruses, Coronaviruses, respiratory syncytial viruses). Sometimes we are at risk for more virulent upper respiratory viruses such as COVID-19. If we are wise, we try to take better care of ourselves during cold and flu season by eating right, getting enough rest, and taking “health and immune-booster” nutrients. It is amazing how, and how many, of our common “immune boosting” nutrients interact with the mitochondria-immune system axis.
For many of us, our susceptibility to infection and viral resistance may be linked to our mitochondrial fitness and nutritional status. Healthy mitochondria interact with our innate and adaptive immune systems during pathogen exposure and infection to promote the appropriate immune responses and eradicate the invader. Dysfunctional mitochondria may not be able to properly signal the immune system during an infection, or worse, may inappropriately send chronic pro-inflammatory signals that contribute to inflammation and disease.
Read More: Oxidative Stress, Viral Infection, and Mitochondrial Function—Are Your Mitochondria “Tough Enough?”
Many of our go-to nutrient immune system boosters work through interaction with our mitochondrial and immune cells as cofactors, antioxidants, cell signals, and cell energy sources.
Cofactors are vitamins and minerals that are incorporated into enzyme proteins or serve as catalysts during enzyme interactions. The mitochondrial-immune system axis has several enzymes with specific cofactor requirements. Antioxidant enzymes, which are used to prevent oxidative stress (ROS) within mitochondria, are also necessary to restore tolerance after an infection. Viruses often induce ROS within infected cells to improve viral replication; therefore, controlling excess ROS is essential for strong viral resistance. Catalase [CAT; heme iron, B3] is the primary defense against cellular ROS, and superoxide dismutase can neutralize superoxide radicals and has both cytoplasmic [SOD1; copper, zinc] and mitochondrial [SOD2; manganese] isoforms.
Antioxidant status is determined not only by the activity of our antioxidant enzymes, but by our dietary antioxidant intake and our innate ability to synthesize and recycle glutathione (GSH).
Glutathione is required to quench ROS and activate antioxidant vitamins C and E. GSH is synthesized on a magnesium-dependent pathway, and GSH depletion inhibits immune cell functions. Once oxidized, GSH can be reduced and re-used by the cell via glutathione reductase [GSR; B2, B3].
Selenium is required by glutathione peroxidases, which protect heme and other host proteins from ROS. Of note, selenium improves mitochondrial respiration and Th1 immune cell responses. Supplements using selenocysteine, or food sources of selenium, may improve assimilation into cells.
N-acetylcysteine and B6 may be supplemented to improve glutathione synthesis in those with low GSH levels or high GSH demand. B6 also supports immune cell proliferation.
Vitamin C is imported into mitochondria to maintain redox status. Vitamin C supports immune cell proliferation and differentiation and is also sequestered into phagocytic immune cells to improve pathogen removal.
Vitamin E improves T-cell function and protects immune cell membranes from oxidative damage.
Oral antioxidants such as curcumin, gingko biloba, melatonin, etc. may also be taken during viral illness to inhibit viral ROS generation and reproduction. Continued use of oral antioxidants can restore tolerance and prevent unnecessary inflammation after an infection.
Signaling proteins are required by the mitochondrial antiviral signaling protein (MAVS) pathway. MAVS activates pro-inflammatory signal pathways during the acute stages of infection to eradicate the pathogen. Two of the proteins on the MAVs pathway are zinc-dependant. Zinc is also required for antibody production and immune cell differentiation.
Another protein is induced by retinoic acid (vitamin A). Vitamin A is also required for antibody production.
Vitamin D binds directly to vitamin D receptors and response elements within our mitochondria to modulate DNA and RNA expression.
Read More: How Vitamin D Protects Against Respiratory Viruses
Cellular energy source preferences change dramatically when immune cells are activated by MAVS or a pathogen exposure. As naïve immune cells differentiate and become specialized, they may use glucose, glutamine, fatty acids or oxygen respiration (OXPHOS) as their primary energy source:
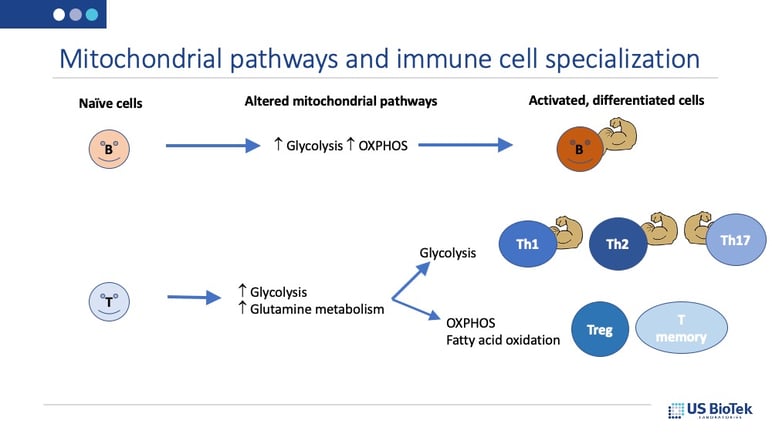
While these common “health and immune booster” nutrients can help us during cold and flu season, they may also be useful year-round as part of a targeted mitochondrial support strategy. Our mitochondrial function and antioxidant status is determined in part by our genetic inheritance, and some of us may need more, and different, nutritional support than others to maintain healthy mitochondrial function and biochemistry. The only way to know for sure which nutrients are needed is to test; it’s not too late to check on mitochondrial function and start supporting these vital immunity-supporting biochemical pathways using a dried urine organic acids profile. The profile results provide targeted nutrient recommendations for truly personalized medicine that can improve mitochondrial and immune function against viruses and other pathogens.
Supporting ourselves with proper diet, rest and nutrition is the best way to weather cold and flu season and prevent upper respiratory infections such as COVID-19. Knowing which nutrients to use, and how they work, empowers us to take charge of our own and our patients’ health.
Angajala A, Lim S, Phillips JB, et al. Diverse Roles of Mitochondria in Immune Responses: Novel Insights Into Immuno-Metabolism. Front Immunol. 2018;9:1605.
Avery JC, Hoffmann PR. Selenium, Selenoproteins, and Immunity. Nutrients. 2018;10(9):1203.
Bermingham EN, Hesketh JE, Sinclair BR, Koolaard JP, Roy NC. Selenium-enriched foods are more effective at increasing glutathione peroxidase (GPx) activity compared with selenomethionine: a meta-analysis. Nutrients. 2014;6(10):4002–4031.
Brison E, Jacomy H, Desforges M, Talbot PJ. Novel treatment with neuroprotective and antiviral properties against a neuroinvasive human respiratory virus. J Virol. 2014;88(3):1548–1563.
Carr AC, Maggini S. Vitamin C and Immune Function. Nutrients. 2017;9(11):1211.
Dato S, Crocco P, D'Aquila P, et al. Exploring the role of genetic variability and lifestyle in oxidative stress response for healthy aging and longevity. Int J Mol Sci. 2013;14(8):16443–16472.
Drake V, Gombart A. Immunity in Depth. Linus Pauling Institute, Oregon State University. https://lpi.oregonstate.edu/mic/health-disease/immunity Accessed 25 march 2020.
Fehr AR, Perlman S. Coronaviruses: an overview of their replication and pathogenesis. Methods Mol Biol. 2015;1282:1–23.
Greiller CL, Martineau AR. Modulation of the immune response to respiratory viruses by vitamin D. Nutrients. 2015;7(6):4240–4270.
Higdon J, Tsuji P. Selenium. Linus Pauling Institute, Oregon State University. https://lpi.oregonstate.edu/mic/minerals/selenium Accessed 25 March 2020.
Liguori I, Russo G, Curcio F, et al. Oxidative stress, aging, and diseases. Clin Interv Aging. 2018;13:757–772.
Lu SC. Glutathione synthesis. Biochim Biophys Acta. 2013;1830(5):3143–3153.
McGuire PJ. Mitochondrial Dysfunction and the Aging Immune System. Biology (Basel). 2019;8(2):26.
Muñoz-Montesino C, Roa FJ, Peña E, et al. Mitochondrial ascorbic acid transport is mediated by a low-affinity form of the sodium-coupled ascorbic acid transporter-2. Free Radic Biol Med. 2014;70:241–254.
Picard M, McEwen BS. Psychological Stress and Mitochondria: A Systematic Review. Psychosom Med. 2018;80(2):141–153.
Poltronieri P, Sun B, Mallardo M. RNA Viruses: RNA Roles in Pathogenesis, Coreplication and Viral Load. Curr Genomics. 2015;16(5):327–335.
Reshi ML, Su YC, Hong JR. RNA Viruses: ROS-Mediated Cell Death. Int J Cell Biol. 2014;2014:467452.
Ricca C, Aillon A, Bergandi L, Alotto D, Castagnoli C, Silvagno F. Vitamin D Receptor Is Necessary for Mitochondrial Function and Cell Health. Int J Mol Sci. 2018;19(6):1672.

What is GI transit time? Gastrointestinal (GI) transit time is the duration required for ingested material to pass through the digestive tract from...
Overview Many clinicians searching for answers about mycotoxin testing ask questions like “Should I provoke mycotoxins before urine testing?”, “When...

US BioTek Laboratories today announced the completion of its merger with NutriPATH Pathology, a respected functional pathology laboratory serving...