GIT Transit Time & SIBO: Accurate Testing Starts with Timing
What is GI transit time? Gastrointestinal (GI) transit time is the duration required for ingested material to pass through the digestive tract from...
3 min read
![]() Dr. Andrea Gruszecki, ND
:
March 17, 2020 at 3:18 PM
Dr. Andrea Gruszecki, ND
:
March 17, 2020 at 3:18 PM

Every year we run the risk of viral upper respiratory infection by RNA viruses including influenza and common colds (Rhinoviruses, Coronaviruses, respiratory syncytial viruses). Sometimes we are at risk for more virulent upper respiratory viruses such as COVID-19.
For many of us, our susceptibility to infection and viral resistance may be linked to our mitochondrial fitness and antioxidant status. Unfortunately, if we don’t check, we don’t know how our mitochondria are functioning, or if they are “tough enough” to protect us if we are exposed to a pathogen. Our mitochondrial function and antioxidant status is determined in part by our genetic inheritance, however, there are also environmental factors that impact mitochondrial health and antioxidant levels such as nutrient levels, age, comorbid illness, toxic exposures and stress.
Mitochondrial function and immune function are tightly linked. Of the six primary metabolic pathways that affect immune function, five of these processes take place in the mitochondria: the citric acid cycle, fatty acid oxidation, electron transport chain (oxidative phosphorylation or OXPHOS), calcium buffering, and heme biosynthesis.
Exposure to a pathogen, such as a respiratory virus, alters the use of several of these biochemical pathways, including the citric acid cycle, OXPHOS, and fatty acid oxidation in the immune cell mitochondria. Each of these alterations has specific effects in programming immune responses, and the pathways that are altered varies with the type of immune cell:
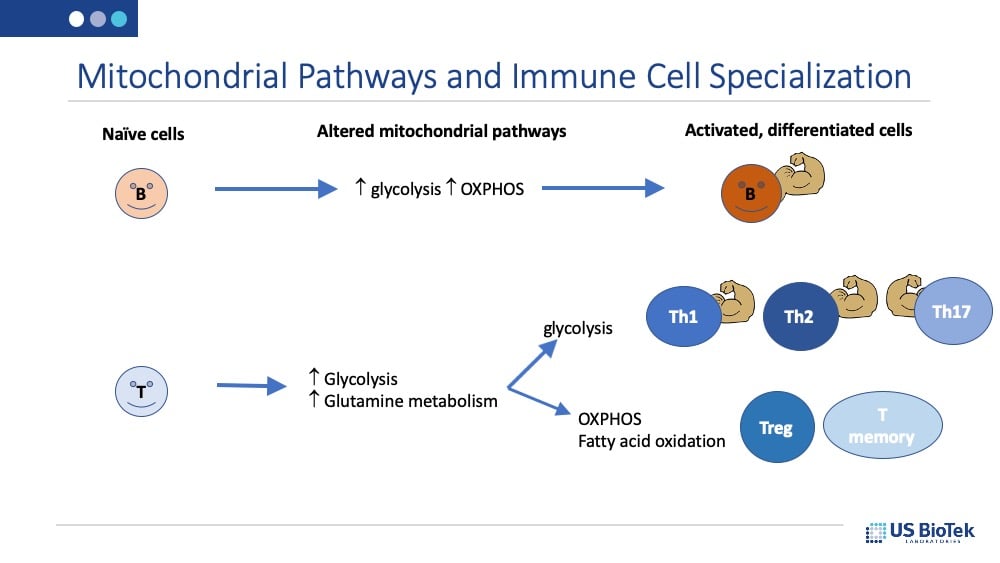
In addition, the presence and metabolism of specific amino acids, such as glutamine and tryptophan, also promote immune responses. Glutamine is required for T-cell differentiation and is converted into alpha-ketoglutarate to support the citric acid cycle. A lack of glutamine inhibits oxygen consumption and ATP production in effector T-cells. Tryptophan deficiency inhibits T-cell proliferation, while some peripheral metabolites of tryptophan, such as kynurenine, may have tolerance-promoting properties. It is also possible for excess tryptophan to cause imbalances in kynurenine metabolism that, combined with a dysregulated Th1/Th2 immune profile, may induce oxidative stress.
Due to the presence of the electron transport chain in the mitochondria, they are a primary source of reactive oxygen species (ROS) in the cell. Mitochondrial ROS (mtROS) are vital for immune system signaling to regulate DNA transcription in immune cells, and to stimulate protective pro-inflammatory responses during pathogen exposure and the acute stages of infection. mtROS modification of signaling pathways induces nuclear factor kappa light chain enhancer of activated B-cells (NF-kB). During a pathogen exposure, acute inflammatory responses help us resist infection. Cellular signals, such as NF-kB and mtROS together induce anti-viral inflammasome pathways, a vital anti-viral pathway in our innate immunity. NF-kB, in conjunction with other mitochondrial signals, induces the expression of protective type I interferons to fight the infection.
However, once a viral pathogen has established itself, ROS production may become problematic, as many RNA viruses deliberately increase cellular oxidative stress to improve viral replication and suppress immune responses. And here’s where we can run into problems: if we lose our antioxidant capacity, we lose our ability to quench ROS production and our inflammatory anti-viral responses when the infection is over. Even worse, we may be more susceptible to infections if we have poor mitochondrial function and poor antioxidant status!
Our mitochondrial function and antioxidant status is determined in part by our genetic inheritance, however there are also environmental factors that impact mitochondrial health and antioxidant levels:
While there are no pathogen-specific changes, human studies of urinary organic acids show that viral infections dysregulate the citric acid cycle and other mitochondrial functions. If our mitochondria are dysregulated prior to a pathogen exposure, we are more susceptible to infection, and more likely to suffer severe infections, than someone with well-nourished mitochondria and functional biochemical pathways.
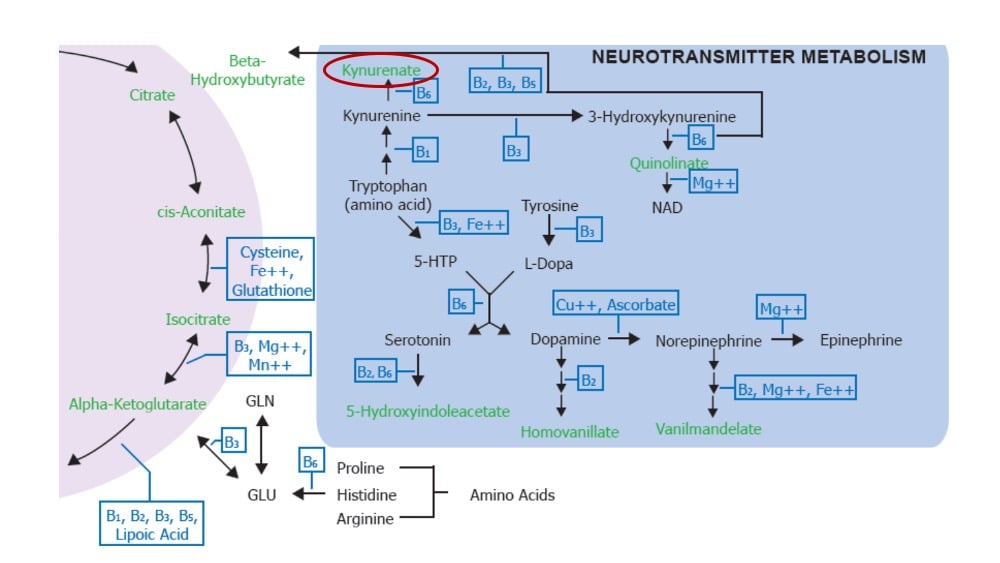
It’s not to late to check on mitochondrial function and start supporting these vital immunity-supporting biochemical pathways using the dried urine Organic Acids profile. The profile results provide targeted nutrient recommendations for truly personalized medicine that can help “toughen up” mitochondrial and immune function against viruses and other pathogens.
Angajala A, Lim S, Phillips JB, et al. Diverse Roles of Mitochondria in Immune Responses: Novel Insights Into Immuno-Metabolism. Front Immunol. 2018;9:1605.
Chappel, B. (2020) Coronavirus: COVID-19 Is Now Officially A Pandemic, WHO Says https://www.npr.org/sections/goatsandsoda/2020/03/11/814474930/coronavirus-covid-19-is-now-officially-a-pandemic-who-says Accessed 11 March 2020.
Dato S, Crocco P, D'Aquila P, et al. Exploring the role of genetic variability and lifestyle in oxidative stress response for healthy aging and longevity. Int J Mol Sci. 2013;14(8):16443–16472.
Fehr AR, Perlman S. Coronaviruses: an overview of their replication and pathogenesis. Methods Mol Biol. 2015;1282:1–23.
Liguori I, Russo G, Curcio F, et al. Oxidative stress, aging, and diseases. Clin Interv Aging. 2018;13:757–772.
Mándi Y, Vécsei L. The kynurenine system and immunoregulation. J Neural Transm (Vienna). 2012;119(2):197–209.
McGuire PJ. Mitochondrial Dysfunction and the Aging Immune System. Biology (Basel). 2019;8(2):26.
Mellor AL, Munn DH. Tryptophan catabolism and regulation of adaptive immunity. J Immunol. 2003;170(12):5809–5813.
Pesce M, Sergi MR, Rizzuto A, et al. Associations between the antioxidant network and emotional intelligence: a preliminary study. PLoS One. 2014;9(7):e101247.
Picard M, McEwen BS. Psychological Stress and Mitochondria: A Systematic Review. Psychosom Med. 2018;80(2):141–153.

What is GI transit time? Gastrointestinal (GI) transit time is the duration required for ingested material to pass through the digestive tract from...
Overview Many clinicians searching for answers about mycotoxin testing ask questions like “Should I provoke mycotoxins before urine testing?”, “When...

US BioTek Laboratories today announced the completion of its merger with NutriPATH Pathology, a respected functional pathology laboratory serving...