To Provoke, or Not To Provoke?: Mycotoxin Provocation Testing
Overview Many clinicians searching for answers about mycotoxin testing ask questions like “Should I provoke mycotoxins before urine testing?”, “When...
6 min read
![]() Dr. Andrea Gruszecki, ND
:
September 21, 2020 at 7:20 PM
Dr. Andrea Gruszecki, ND
:
September 21, 2020 at 7:20 PM

Polluted air irritates lung tissue and disrupts the lung mucosal barrier. Much of the Pacific coast of the United States is currently impacted by the smoke and destruction of wildfires, and that smoke will eventually circle the earth. While the effects of air pollution on humans is well-documented, the effects of air pollution and other environmental stressors on plants is not commonly known. Convincing evidence now indicates that air pollution can decrease the microbiome diversity of pollen microbiomes and increase the allergenicity of plant pollens. This increased allergenicity, combined with the longer pollen seasons induced by climate change, can mean extra weeks of misery for those with seasonal allergies. The combined inflammatory effect may potentially exacerbate asthma and systemic allergies, as well as increase the risk of pathogen-driven diseases such as COVID-19.
Everything in the natural world likely has an associated microbiome, a collection of bacteria, fungi, and viruses that can influence the health and disease of living organisms through molecular signaling with the host. Microbes found on plant pollens and microbes found in dust, sand and air pollution particulates interact with the human immune system in two different ways; they can trigger immune responses at the lung mucosal barrier (or deeper in the lung tissue if the barrier is compromised) or the pollens and microbes can be swallowed and interact with the gastrointestinal microbiome and the gut mucosal barrier. The majority of microbes in aero-microbiomes are soil bacteria, however air particulates may also contain pathogens such as Streptococcus pneumoniae and fungi such as Aspergillus fumigatus known to cause human disease.
Major air pollution sources include fossil fuel use, industry, agriculture, vehicles, and wildfires. Over 90% of the world’s population lives in air so polluted that it does not meet the World Health Organization (WHO) guidelines. WHO guidelines describe limits in terms of particulate matter size and concentration in air. Toxic gases such as nitrogen oxides can become fine particulates over time. In addition, particulates in air pollution may contain toxic metals that can increase oxidative stress and alter immune functions. Airborne particulates can also support their own microbial microbiomes and may carry some lung pathogens. Any particulate matter (PM) less than 10 µm (PM10) can cause eye irritation, allergies, or nasal irritation (rhinitis). Smaller particles can penetrate deeper into the lung tissues. Human studies indicate that chronic exposure to PM10 is associated with increased hospitalization for respiratory problems. PM2.5 have been associated with an increased risk of lung cancer, stroke, heart disease, neuroinflammation and neurodegeneration. Human studies have shown that exposure to air pollution increases lung inflammation and the risk of COVID-19 infection.
Studies indicate that air pollution exposure in two plants with known allergic potential, birch (Betula species) and timothy grass (Phleum pratense), increases the expression of plant proteins that induce allergic reactions. Human skin-prick tests with a water-based pollen solution indicates that birch pollen from high ozone environments create larger skin wheals and flares than birch pollen from low-ozone areas.
In addition, the microbial diversity on birch and timothy grass pollens has been correlated with onsite air pollution levels including ozone and nitrogen dioxide (NO2). Birch exposed to urban ozone and NO2 air pollution and other environmental stressors produce pollen grains higher in the clinically important antigen Bet v 1. Pollen from stressed birch also have an altered profile of pollen-associated lipid mediators (PALMs) and decreased pollen microbiome diversity which can decrease immune system tolerance to pollens. While timothy grass pollen microbiomes were not significantly different in urban and rural areas, there were significant associations with PALM profiles and losses of microbiome diversity in more urbanized areas.
Sensitization rates to pollen can average 20-30%, and food allergies are increasingly common in the general population. Altered antigen profiles in pollens may increase the likelihood of food-pollen cross-reactions, such as hazelnut and birch. Aeroallergen pollen reactions are classically mediated by IgE or stimulated by Bet v 1-specific T-cells. However, studies of lung fluid in patients with asthma or other respiratory conditions reported higher levels of antigen-specific IgG1 and IgG4 along with IgE. This means that allergy and sensitivity to aeroallergens can be monitored by either serum (IgE, IgG, IgG4) or dried blood-spot testing (IgG, IgG4).
Up to 60% of total IgG is IgG1. This means that dried blood-spot (DBS) samples, which can measure IgG (class 1,2,3) and IgG4 can be used to screen for aeroallergens. IgG1-3 activates the proinflammatory complement pathway and contributes to chronic inflammation. If only a DBS sample is used, it is important to measure IgG4 separately from the other IgG classes because IgG4 has a different function than the other IgG classes.
IgG4 is a blocking antibody for IgE; it binds to IgE receptors and prevents allergic reactions. Measuring IgG4 separately from IgG 1-3 provides the clinician with a great deal of information about chronic inflammatory aeroallergen responses (IgG 1-3) and about IgE reactivity (IgG4). The additional measurement of serum IgA can provide additional insight, as serum IgA can reflect the activity of secretory IgA (sIgA) at gut and nasal mucosal barriers, which is supposed to bind to pathogens and antigens and prevent them from crossing the mucosal barrier into circulation. The example below shows that, for this particular patient IgE-mediated allergies (IgG4) are not a likely cause of the patient’s seasonal and indoor aeroallergen reactions, but that the reactions are mediated by pro-inflammatory IgG 1-3 and IgA-mediated interactions at mucosal barriers.
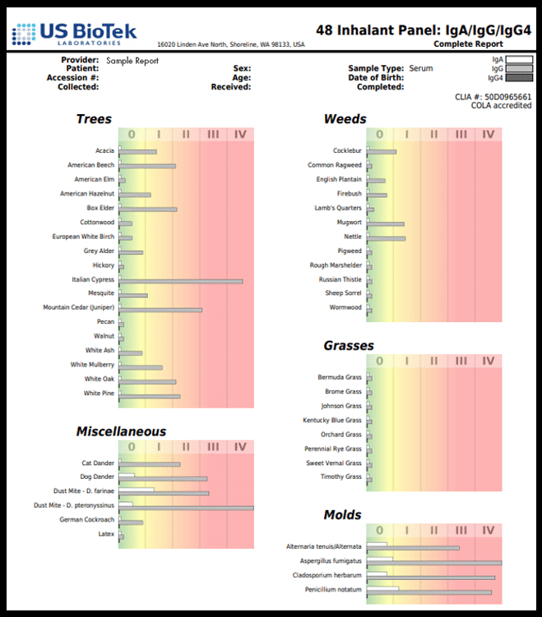
People living in polluted environments are more likely to experience aeroallergen reactions, which may be mediated either by IgE allergy or IgG-mediated chronic inflammation. Those at greatest risk include children and individuals with pre-existing lung or heart disease or other chronic inflammatory disorders. Individuals exposed to the severe wildfire smoke moving across the United States at present may experience new aeroallergen reactions or have reactions for the first time. Testing all patients with food or aeroallergen-related symptoms may provide new information and alter patient management to improve lung function and air quality for these individuals.
Learn more about environmental pollutants on food sensitivities, allergies, and the microbiome.
Overview Many clinicians searching for answers about mycotoxin testing ask questions like “Should I provoke mycotoxins before urine testing?”, “When...

US BioTek Laboratories today announced the completion of its merger with NutriPATH Pathology, a respected functional pathology laboratory serving...

Unlocking the Secrets of Cellular Energy